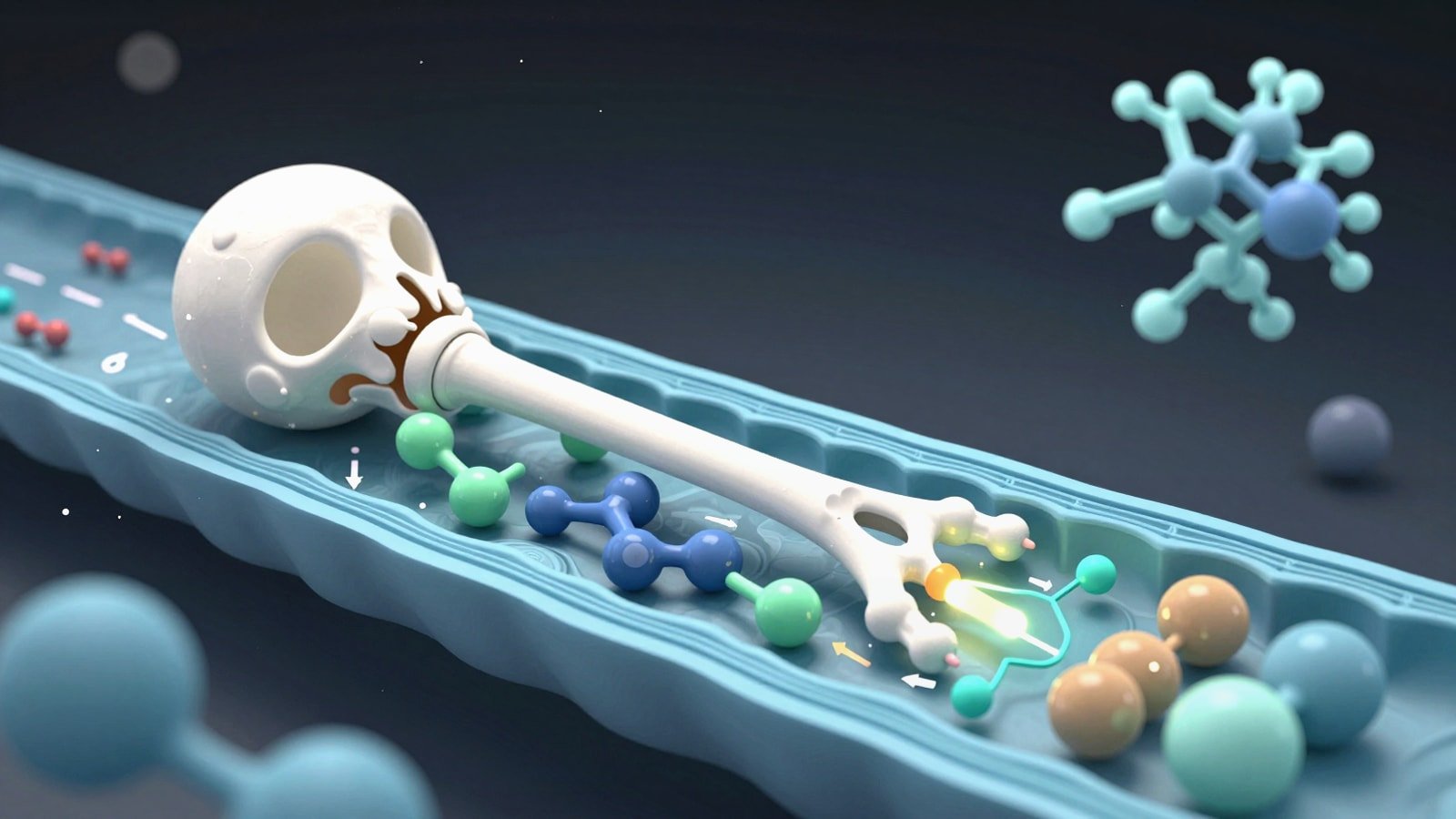
Why Base Editing Demands Better Animation
A strong base editing mechanism of action animation has a harder job than many other therapeutic visuals. It must show molecular targeting, enzyme chemistry, editing windows, DNA repair bias and delivery context in one coherent sequence. If any one of those layers is vague the audience walks away with the wrong mental model. They may think the system cuts both DNA strands like standard CRISPR nuclease editing or that every guide-bound base is changed with perfect selectivity.
Base editing is more subtle than cut-and-repair gene editing. Cytosine base editors and adenine base editors change one base into another without creating a programmed double-strand break. That difference is the center of the story. Your animation should therefore emphasize precision chemistry over brute-force cleavage. The viewer needs to understand that the editor is a guided catalytic machine with a narrow activity window rather than a simple pair of molecular scissors.
For biotech teams this matters in investor decks, clinical communications and platform storytelling. A clean base editing animation can explain why a program targets a point mutation, why the liver is the organ of interest and why lipid nanoparticle delivery changes the therapeutic path. If your team also builds technical scenes in Blender it helps to review our guide to molecular nodes protein scenes before blocking the sequence.
- Make the no-double-strand-break concept visually explicit early
- Treat chemistry and delivery as separate story beats
- Use camera language to distinguish targeting from editing outcome
The Core Scientific Story Your Animation Must Preserve

The scientific backbone of a CRISPR base editor visualization is straightforward when reduced to essentials. A guide RNA brings a Cas-derived targeting module to a genomic locus. The DNA opens into an R-loop that exposes a short single-stranded segment. A tethered deaminase acts on bases within a limited editing window. Cellular repair and replication then convert that temporary chemical change into a durable sequence outcome.
For cytosine base editing the key event is deamination of cytosine to uracil within DNA. In practical storytelling terms the viewer should see C become U first and only later resolve to a stable T after repair or replication logic plays out. For adenine base editing the catalytic event is deamination of adenine to inosine which the cell reads like guanine. In animation this means you should avoid jumping directly from A to G with no mechanistic intermediate unless the audience is very general and the simplification is stated elsewhere.
It also helps to show why Cas9 nickase variants are commonly used. The nick on the non-edited strand biases repair toward installation of the intended edit. That bias is not a guarantee of perfect outcomes yet it is an important reason why editor architecture matters. If your scene treats every edit as automatic you lose the logic that separates first-generation constructs from more optimized editor systems.
- Guide RNA locates the site
- R-loop exposure creates the editable single-stranded zone
- Deaminase chemistry happens before final base conversion is fixed
| Editor class | Catalytic change shown in animation | Typical durable outcome |
|---|---|---|
| Cytosine base editor | C to U | C:G to T:A |
| Adenine base editor | A to I | A:T to G:C |
How to Structure a Clear Base Editing Animation Sequence

The best base editing animation is usually a seven-step sequence rather than one continuous blur of motion. Start with delivery context and cell targeting. Then cut to cell entry and cargo release. Next establish the editor complex with labels for guide RNA, Cas nickase and deaminase. After that move into genomic docking, strand opening and editing-window focus. Only then should you show chemistry, repair bias and final sequence outcome.
This sequence works because each shot answers one question. Where does the therapy go. What enters the cell. How does the complex find DNA. Which base is changed. Why does the edit persist. When teams skip these transitions the animation becomes technically dense but cognitively weak. The audience sees molecules moving yet cannot explain the causal chain.
From a production standpoint this is also the easiest way to keep one master animation adaptable across use cases. The early shots can serve a platform overview. The middle shots can be reused for a disease program deck. The late shots can be swapped to show a different target base, organ or outcome. That modularity is valuable if you later need companion assets like grant figures or static panels. For that workflow see our post on NIH grant figures.
- Shot 1: organ or tissue targeting
- Shot 2: intracellular release of editor payload
- Shot 3: editor complex assembly
- Shot 4: genomic recognition
- Shot 5: base conversion chemistry
- Shot 6: repair bias and fixation
- Shot 7: protein or phenotype consequence
Visualizing Cytosine and Adenine Editors Without Confusion

Many teams need one visual language that can support both cytosine and adenine programs. That is possible if the animation system keeps the shared scaffold stable and changes only the chemistry layer. The targeting complex, guide RNA logic and editing window can look consistent across programs. What changes is the substrate base, the catalytic label and the downstream base-pair outcome.
For a cytosine editor show the deaminase acting on the exposed cytosine within the activity window. If a uracil glycosylase inhibitor is part of the design it should appear as a supporting component that blocks reversal of the uracil intermediate. This does not need a long digression yet one short labeled beat helps advanced audiences understand why the pathway favors product formation.
For an adenine editor focus on A-to-I conversion and on the fact that inosine is interpreted as guanine. Many viewers do not know that intermediate so skipping it can make the system seem chemically magical. A clean way to solve this is a zoomed annotation panel that briefly shows A becoming I and then I pairing like G during fixation. This keeps the main camera elegant while preserving mechanistic credibility.
- Keep shared architecture constant across editor classes
- Change only the chemistry callout and final base-pair outcome
- Use one zoomed inset to explain U or I intermediates
Building an LNP Gene Editing Animation for In Vivo Programs
An LNP gene editing animation should not treat delivery as a decorative prelude. For in vivo base editing MOA stories delivery is part of the mechanism because tissue access determines where editing can happen. If your therapy uses lipid nanoparticles to deliver base editor mRNA and guide RNA to the liver then the animation should make that route explicit from the start.
A practical sequence begins with intravenous administration followed by circulation and liver tropism. Then show hepatocyte uptake, endosomal entry and endosomal escape. After release into the cytosol the mRNA is translated into the base editor protein while the guide RNA becomes available for complex formation. This is the right place to distinguish cargo types. The viewer should understand that the LNP carries instructions and guide components rather than a preformed molecular machine in most current mRNA-based designs.
This distinction is especially important in platform storytelling because it clarifies why transient expression can be beneficial. The payload is delivered, translated and then cleared over time while the genomic edit remains. That is a very different message from persistent viral expression. If you need inspiration for keeping multi-step biology readable across crowded pathways our article on bispecific antibody mechanism of action animation covers similar clarity problems in another complex modality.
- Show organ targeting before molecular editing
- Differentiate mRNA cargo from guide RNA cargo
- Include endosomal escape if delivery performance is a key claim
| LNP story beat | What viewers should learn |
|---|---|
| Systemic administration | Therapy is delivered in vivo rather than ex vivo |
| Liver uptake | Target organ access is intentional |
| Endosomal escape | Cargo must reach the cytosol |
| mRNA translation | Base editor protein is produced inside the cell |
| RNP assembly and nuclear access | Editing machinery becomes functional at the target site |
Common Mistakes in CRISPR Base Editor Visualization
The most common mistake is showing a dramatic DNA cut because it feels familiar and cinematic. That immediately undermines the point of base editing. If cleavage must be referenced at all it should be framed as what conventional nuclease editing does not what the base editor is primarily designed to do.
A second mistake is hiding the editing window. Base editing is not a target-any-base tool. Sequence context, PAM access and positional editing windows shape what can be changed. You do not need to drown the viewer in numbering yet the animation should signal that only a narrow zone inside the protospacer is catalytically active.
A third mistake is conflating molecular success with therapeutic success. Even if the editor makes the intended substitution the audience still needs one final bridge to phenotype. Show reduced toxic protein production, restored protein function or durable knockdown of a liver-secreted factor. Otherwise the story ends at the genome and leaves business or clinical audiences asking why they should care. This problem also appears in cell therapy storytelling and we address it in our CAR-T mechanism of action animation guide.
- Do not depict a double-strand break as the main event
- Do not hide the editing window
- Do not stop the story before the biological consequence
A Production Framework for Biotech Teams
If you are commissioning a base editing mechanism of action animation the most efficient process starts with a science lock before style frames. Define the editor class, cargo format, target tissue, intended nucleotide change and desired biological consequence. Then identify which details are mandatory for experts and which can stay in optional overlays or companion stills.
Next build a script that separates voiceover logic from visual logic. A good script often says less than the visuals show. For example the narration can say that the guide RNA directs the editor to the target sequence while the screen simultaneously labels the PAM, exposes the R-loop and highlights the active window. This prevents clunky over-explanation and keeps the film moving.
Finally create output tiers. One hero animation can support a long homepage version, a conference loop, an investor clip and static derivatives for decks or posters. During that process accessibility should not be an afterthought. Label contrast, color independence and descriptive alt text matter for scientific audiences too. Our post on scientific figure accessibility and alt text is useful when the animation also needs figure derivatives.
- Lock the science before you design the look
- Separate narration from on-screen explanation
- Plan reusable outputs from one molecular master scene
FAQ
What is the difference between a base editing animation and a general CRISPR animation?
AA base editing animation must emphasize single-base chemistry, limited editing windows and the absence of a programmed double-strand break. A general CRISPR animation often centers on nuclease cutting and repair pathways instead.
What should a CRISPR base editor visualization include at minimum?
AIt should include the guide RNA, the Cas-derived targeting module, the deaminase component, the genomic target site, the editable window and the final sequence consequence. For in vivo programs it should also include delivery context such as LNP uptake and intracellular release.
How do you explain in vivo base editing MOA to a non-specialist audience?
AStart with the delivery route and the therapeutic goal. Then show that the editor is guided to one DNA address and changes one letter rather than cutting the chromosome apart. End with the protein or disease effect rather than the nucleotide change alone.
Do all LNP gene editing animations need to show endosomal escape?
ANot always. If the piece is a short high-level brand film you may imply intracellular release. If the program story depends on delivery performance, tissue selectivity or platform differentiation then endosomal escape is worth showing explicitly.
How long should a base editing mechanism of action animation be?
AFor most biotech marketing and BD use cases 60 to 120 seconds is enough for a hero version. More detailed training or investor assets can extend beyond that if the sequence is modular and each beat answers a clear question.
CTA
If your team needs a base editing animation that is scientifically precise and commercially useful we design mechanism of action visuals for complex therapeutic stories from first script through final render. We can help you simplify editor architecture, build a convincing LNP gene editing animation and adapt the same molecular scene for web, conference and investor use.
Explore the process on our homepage or contact us to discuss a base editing mechanism of action animation tailored to your platform, program and audience.